Advertising Information
- Kidney Cancer Journal is the only peer-reviewed medical journal and most comprehensive source of scientific information focusing exclusively on renal cell carcinoma
- Robert A. Figlin, MD, FACP , Editor-in-Chief. Principal investigator on numerous kidney cancer clinical trials. Dr. Figlin, MD is the Associate Director of the Academic Development Program and Director of the Division of Hematology/Oncology at Cedars-Sinai’s Samuel Oschin Comprehensive Cancer Institute. He is board-certified in both medical oncology and internal medicine.
- Dr. Senthil Samy, PhD
Executive Editor and Publisher.
Contact:
sen'at'kidney-cancer-journal.com | Phone:+1 (919) 694-3599 - Published quarterly since 2003
- Distributed as a complimentary professional service to approximately 12,300 physicians, including all medical oncologists and targeted urologists.
- Utilizes a Medical Advisory Board and Editorial Advisory Board of more than 30 physicians who are leading investigators in this field.
- Archives available through the KCA’s website at www.kidneycancer.org
- Special Reports and supplements covering ASCO and highlights from the annual International Kidney Cancer Association Meeting
* Program pricing available upon request
- For Sponsored contents and clinical strategies: Contact: sen'at'kidney-cancer-journal.com
Patient Outreach, Engagement, Advocacy, and Support
The Kidney Cancer Association (KCA) is a charitable organization made up of patients, family members, physicians, researchers, and other health professionals. We fund, promote, and collaborate with the National Cancer Institute (NCI), American Society for Clinical Oncology (ASCO), American Urologic Association (AUA), and other institutions on research projects. They educate families and physicians, and serve as an advocate on behalf of patients at the state and federal levels.
The Kidney Cancer Association was founded in 1990 by a small group of patients, including Eugene P. Schonfeld, Ph.D., and medical doctors in Chicago, Illinois. It is a nonprofit charity incorporated in the State of Illinois. It has also been designated as a tax exempt organization under Section 501(c)(3) of the U.S. Internal Revenue Service code. Donations to the Association are tax deductible.
The Kidney Cancer Association is a member of, and is certified by, the National Health Council as an organization in full compliance with the Standards of Excellence for voluntary health agencies.
The Kidney Cancer Association is developing a global presence and is creating websites in several languages, including Chinese, Japanese, Spanish, German, French, Italian and other languages. New patient support groups are planned for the European Union, Asia and Australia in 2007.
Connect with Oncology and Urology Professionals
Digital, Print, Multi-Media, Special Projects
Communications Opportunities
KCJ Digital Solutions Programs
For specific information on these programs contact: Susan Hirschhaut Director of Advertising and Strategic Initiatives susan'at'kidney-cancer-journal.com (847) 476-0588
Circulation TOTAL = 12,300 Physicians
| Medical Oncologists | Hem / Oncologists | Oncologist TKI prescribers | Urologists | Other Oncologists | |
|---|---|---|---|---|---|
| Number | 5,139 | 2,526 | 2,184 | 1,728 | 1,280 |
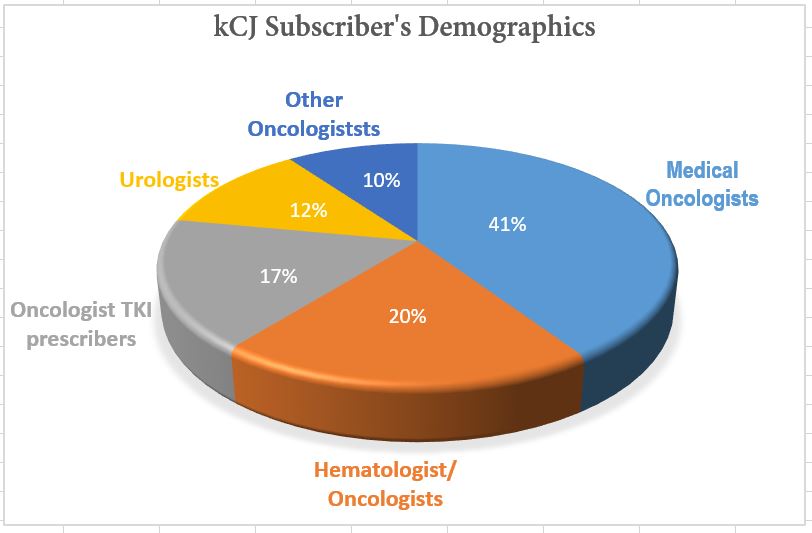
This breakdown is targeted base on ICD10 codes for physicians with a focus on kidney cancer (List provided by DMD/Direct Medical Data, LLC.)
| 1X | 4X | 8X | 16X | 12X | 24X | |
|---|---|---|---|---|---|---|
| Full Page (B&W) | 8,900 | 8,700 | 8,500 | 8,300 | 7,900 | 8,000 |
| 1/2 Page (B&W) | 7,200 | 7,000 | 6,650 | 5,900 | 5,300 | 5,000 |
| 1/4 Page (B&W) | 4,600 | 4,500 | 4,200 | 4,100 | 3,900 | 3,800 |
Color Charges
2 Color $800
4 Color $1,700
Premium Position – Cover 4
A 50% premium charge will be applied to the B&W Full Page rate. Based on the client’s Frequency Rate.
Premium Position – Cover 2
A 25% premium charge will be applied to the B&W Full Page rate. Based on the client’s Frequency Rate.
Mechanical Requirements
Trim Size: 8 3/8” x 10 7/8”| Non-Bleed | Bleed | Bleed Safety | Trim | |
|---|---|---|---|---|
| Ad Size* | (w x h) | (w x h) | (w x h) | (w x h) |
| Spread | 16 1/4” x 10 3/8” | 17” x 11 1/8” | 16 1/4” x 10 3/8’ | 16 ¾” x 10 7/8” |
| Full Page | 7 7/8” x 10 3/8” | 8 5/8” x 11 1/8” | 7 7/8” x 10 3/8” | 8 3/8” x 10 7/8” |
| 1/2 Page Vertical | 3 3/4” x 10 3/8” | 4 3/8” x 11 1/8” | 3 5/8” x 10 3/8” | 4 1/8” x 1- 7/8” |
| 1/2 Page Horizontal | 7 3/4” x 5” | 8 5/8” x 5 5/8” | 7 3/4” x 4 7/8” | 8 3/8” x 5 3/8” |
| 1/2 Page Hor. Spr. | 16 1/4” x 5” | 17” x 5 5/8” | 16 1/4” x 4 7/8” | 16 3/4” x 5 3/8” |
| 1/4 Page | 3 3/4” x 5” | 4 3/8” x 5 5/8” | 3 5/8” x 4 7/8” | 4 1/8” x 5 3/8” |
* Other size ads are available upon request.
| Binding Method: Saddle Stitched | Color: 4 Color (CMYK) |
| Bleed: 1/8 “ (8.675” x 11.125”) | Live Area: 7 “ x 10” |
| Maximum Density: 300 | Safety: Keep Live Matter from trim – 1/4” |
| Spreads: Live area is 15 1/2” x 10” |
Please send all files as press-quality PDF files, with crop marks, bleeds, and a color proof or matching with final press proofs.
Website Advertising Specifications
- 300 x 250 pixels (medium rectangle)
- 468 x 60 pixels (full banner)
- 728 x 90 pixels (leader board)
- 300 x 600 pixels (half-page skyscraper)
- 300 x 100 pixels (low rectangle)
2025 and 2026 Issues and Closing Dates
| Volume 22, Number 1: | February - GU ASCO edition MATERIALS: January 24 |
|---|---|
| Volume 22, Number 2: | June MATERIALS: May 11 |
| Volume 22, Number 3: | September MATERIALS: Aug 28 |
| Volume 22, Number 4: | December MATERIALS: Nov.30 |
For ROB ads, Inserts, Tip-Ons, etc., to be inserted or printed, all materials
can be sent to the attention of:
Susan Hirschhaut
Director of Advertising and Strategic Initiatives
susan@kidney-cancer-journal.com
(847) 476-0588
Advertising Policy
Our advertising policy is consistent with the principles mentioned in the Recommendations on Publication Ethics Policies for Medical Journals which issued by the World Association of Medical Editors (WAME).
http://www.wame.org/recommendations-on-publication-ethics-policie
<- All advertisements and commercially sponsored publications are independent from editorial decisions. Kidney Cancer Journal does not endorse any product or service marked as an advertisement or promoted by a sponsor in Kidney Cancer Journal publications. Editorial content is not compromised by commercial or financial interests, or by any specific arrangements with advertising clients or sponsors.
- Kidney Cancer Journal reserves the right to decline any type of advertising that is damaging to the brand of Kidney Cancer Journal or is inappropriate to the content held on the Kidney Cancer Journal
-
Advertisements may not be deceptive or misleading, and must be verifiable. Advertisements should clearly identify the advertiser and the product or service being offered. Exaggerated or extravagantly worded copy will not be allowed. Advertisements will not be accepted if they appear to be indecent or offensive in either text or artwork, or if they relate to content of a personal, racial, ethnic, sexual orientation, or religious nature.
Advertising for the following categories is prohibited:
- Alcohol
- Tobacco
- Weapons, firearms, ammunition
- Fireworks
- Gambling and lottery
- Pornography or related themes
- Political and religious advertisements
- Advertisements that claim to have a “miracle” cure or method
- Advertisements that make unsubstantiated health claims for the products advertised
- Advertisements directed at children
- Once an advertisement has been deployed online, it will be withdrawn from the journal site at any time if the Editor(s)-in-Chief or Publisher request its removal.
- Kidney Cancer Journal will not allow any treatment-specific or drug-specific campaign to be targeted to a specific article(s) or on any page where content relates to the product(s) being advertised. (Advertisers may not link to articles using keywords; they may not target advertising for a specific product on the condition that it appear in the same location and at the same time as a specific article mentioning that product and they may not refer to an article published at the same time as the advertisement appears).
- All advertisements for drug-specific campaigns must comply with the relevant US and Euorpean legislation that regulates advertising. Advertisers should make available to Kidney Cancer Journal the marketing authorization and summary of product characteristics when submitting their advertisement. In the case of drug advertisements, the full generic name of each active ingredient should appear. Each page of an advertisement for a prescription-only medicine should be clearly labeled as intended for health professionals.
- All advertisements for drug specific campaigns should encourage correct and rational use and must not be misleading.
- Advertisements and editorial content must be clearly distinguishable. Kidney Cancer Journal will not publish “advertorial” content, and sponsored supplements must be clearly indicated as such. If a supplement did not undergo peer review or underwent a peer review-process different from the rest of the journal that should be explicitly stated.
- Editorial decisions will not be influenced by current or potential sponsors and advertisers, and will not be influenced by marketing decisions. Advertisers and sponsors have no control or influence over the results of searches a user may conduct on the website by keyword or search topic.
Advertising complaints policy
Please send any complaints about advertising to: info@kidney-cancer-journal.com